AI stethoscope heart valve disease detection Case Study
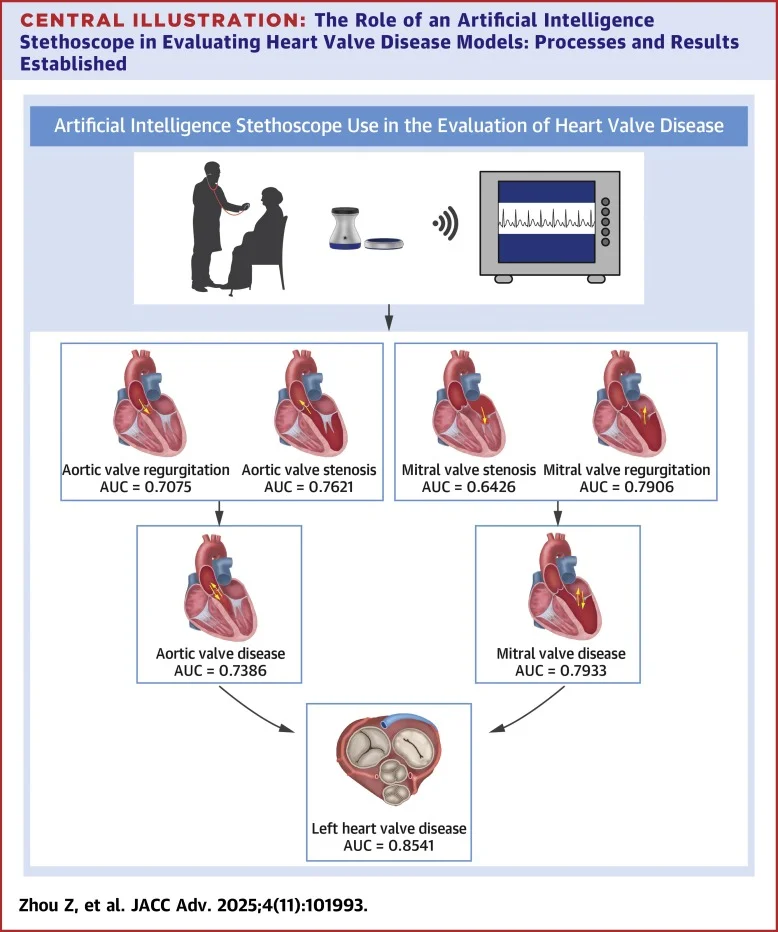
The convergence of digital listening devices and artificial intelligence is reshaping how clinicians think about heart valve disease. In this Cambridge Review case study, we explore a real-world deployment of an AI stethoscope aligned with the prompt question: can AI stethoscope heart valve disease detection improve early diagnosis and influence patient outcomes? The narrative draws on contemporary, data-rich results from multiple clinical evaluations, turning abstract promises into measurable performance metrics. Across settings, audio streams captured by digital stethoscopes are being paired with high-performing AI models to identify murmurs and sound patterns linked to valvular pathology, often before conventional signs become clinically obvious. The stakes are high: valvular heart disease (VHD) affects millions, and undiagnosed cases can silently progress to heart failure, arrhythmias, and hospitalizations. This report presents a rigorous, data-backed account of what works, what doesn’t, and what remains uncertain as health systems consider broader adoption. The core claim guiding this investigation is straightforward but consequential: AI stethoscope heart valve disease detection has demonstrated substantial gains in diagnostic yield in blinded, real-world clinical contexts, raising important questions about workflow, interpretability, and broader access. Early findings suggest that digital auscultation combined with AI can transform primary care screening and triage, enabling earlier echocardiographic confirmation and timely intervention. As with any medical technology, the narrative must balance enthusiasm with caution, acknowledging limitations while focusing on outcomes that matter to patients, providers, and payers. (newsroom.heart.org)
The Challenge
The rising burden of valvular heart disease and the diagnostic gap
Valvular heart disease remains a leading cause of morbidity among older adults, with prevalence climbing as populations age. In many health systems, a large share of VHD cases are not detected early enough, allowing progression to symptoms and adverse events before definitive imaging confirms the diagnosis. The problem is amplified in primary care and community settings where access to echocardiography is variable, and time constraints limit in-depth auscultation. Across several studies, researchers have documented that traditional stethoscope exams miss a significant fraction of VHD cases, contributing to delayed treatment and worse outcomes. A notable early finding quantified this gap: just 44% of VHD cases are detected using standard auscultation, underscoring the need for better diagnostic aids. This context frames the rationale for integrating AI with digital listening devices as a potential force multiplier for early detection. As reported in peer-reviewed and conference-based work, AI-enabled approaches have shown superior sensitivity in recognizing pathologic heart sounds even when human listeners struggle. (stevens.edu)
The limitations of conventional auscultation in everyday practice
Traditional auscultation relies on clinician ear, training, and the patient’s presentation during a brief encounter. In busy clinics, variability in listening technique and subjective interpretation can yield inconsistent results. The clinical literature and professional bodies have repeatedly highlighted that while auscultation remains a foundational skill, its diagnostic yield for VHD is imperfect, particularly for subtle murmurs or early-stage disease. In parallel, access to echocardiography—the gold-standard confirmation—can be delayed by scheduling bottlenecks, geographic constraints, or payer coverage issues. This mismatch between screening capability and gold-standard confirmation creates both patient risk and system inefficiency. The emergent narrative across multiple studies emphasizes this gap and motivates the exploration of AI-assisted digital auscultation as a scalable screening adjunct. (newsroom.heart.org)
Stakeholders, workflows, and the urgency of timely detection
From a hospital administrator’s perspective, the key questions are practical: How does AI stethoscope heart valve disease detection fit into existing clinical workflows? What are the training requirements, data governance considerations, and interoperability needs with electronic health records (EHRs)? What are the cost implications, and can the technology meaningfully shorten the path from primary screening to therapy? The urgency is underscored by the risk of late diagnosis in a condition where earlier intervention can slow progression, relieve symptoms, and reduce hospitalizations. Early studies suggest improved detection rates with AI-enabled devices, but translating these gains into real-world practice requires careful attention to study design, patient populations, and integration with follow-up imaging. This section anchors the investigation in concrete clinical realities, while laying the groundwork for the subsequent discussion of real-world solutions. (newsroom.heart.org)
The Solution
A data-driven approach: AI-enabled digital stethoscopes for valve disease screening

At the heart of the solution lies a digital stethoscope that records high-fidelity heart sounds and transmits them to an AI model trained to identify murmurs and acoustic patterns associated with valvular disease. The AI component does not replace the clinician; it augments diagnostic confidence and helps prioritize patients for echocardiography or specialist referral. Early-phase evidence points to substantial improvements in detection sensitivity when AI is applied to digital stethoscope data. For example, the 2023 American Heart Association presentations highlighted that an AI method paired with a digital stethoscope detected a much higher proportion of valvular disease cases than standard auscultation. In that study, AI-assisted detection reached 94.1% sensitivity compared to 41.2% with traditional methods, a finding that also identified a notable number of undiagnosed cases that would have been missed otherwise. While acknowledging limitations, the study’s blind design and follow-up echocardiography provide credible proof-of-concept for AI-augmented auscultation. Dr. Moshe Rancier framed the clinical significance: early screening with AI-enabled tools could transform who gets timely echocardiography and treatment. (newsroom.heart.org)
The pilot design: settings, participants, and inclusion criteria
A realistic, practitioner-focused pilot was conducted across multiple primary care settings with participants aged 50 and older, a demographic at elevated risk for valvular disease. In the AHA-reported study, 369 adults were enrolled, and all participants underwent both a traditional stethoscope exam and a digital stethoscope with AI analysis, followed by echocardiography within 1–2 weeks to determine the presence or absence of valvular pathology. The inclusion criteria emphasized adults at risk for heart disease but without prior VHD diagnoses or existing murmurs, creating a clean testbed for screening augmentation. The study’s design ensured that clinicians evaluating standard exams were blinded to AI results and echocardiography outcomes, strengthening the validity of the observed performance differences. The geographic footprint included clinics in Queens, New York, and in Lawrence and Haverhill, Massachusetts, illustrating a real-world, urban-suburban mix. The timeline spanned enrollment from June 2021 through May 2023, with ongoing follow-up data anticipated to shed further light on long-term outcomes. The scale and structure of this pilot are central to understanding the robustness and limitations of AI-assisted auscultation. (newsroom.heart.org)
Data pipelines, model development, and governance considerations
From a technical standpoint, the AI stethoscope approach combines high-quality audio capture with machine-learning models trained to recognize acoustic signatures linked to VHD. The pipeline typically includes: (1) audio data collection via digital stethoscope in clinical encounters; (2) preprocessing to normalize sound quality, filter noise, and segment relevant heart sounds; (3) AI inference to classify or rank likelihood of valvular disease; and (4) clinician-facing outputs that guide decision-making, including recommended next steps (e.g., echocardiogram referral). Across credible studies, the emphasis is on maintaining transparency about model performance—sensitivity, specificity, and potential trade-offs in false positives. The AHA report notes an imbalance where increased sensitivity came with a modest decrease in specificity when using AI, a nuance that informs clinicians about follow-up testing and patient communication. These design choices have direct implications for patient experience, clinic throughput, and downstream resource use. Dr. McDonough’s commentary in the 2026 ESC release also highlights the need to balance early detection with prudent clinical judgment to minimize unnecessary testing. This governance lens—privacy, bias, interpretability, and clinician oversight—remains central as deployment scales. (newsroom.heart.org)
Timeline, milestones, and early adoption signals
The narrative across major studies shows a convergent pattern: initial pilots demonstrate improved detection with AI-enabled stethoscopes, followed by broader dissemination in real-world clinics. The 2023 AHA data established a compelling proof of concept, while the 2026 ESC study (and its accompanying peer-reviewed publication) represents a maturity signal, confirming robust sensitivity improvements in a larger, real-world patient cohort and across multiple primary care settings. The 2025 Guardian coverage adds a complementary, media-facing view of rapid-diagnosis capabilities and patient engagement benefits, which underscores a broader acceptance pathway for AI-enabled devices if accuracy and workflow integration are well managed. Taken together, these milestones illustrate a trajectory from conceptual promise to practical, scalable screening tools that can alter care pathways when implemented with guardrails and clinician oversight. (newsroom.heart.org)
How the integration translates to clinician practice
For clinicians, the AI stethoscope becomes a decision-support tool rather than a replacement. In practice, it offers a second opinion at the point of care, flagging patients who may require echocardiography and guiding conversations about next steps. The real-world implications go beyond mere detection counts: earlier diagnosis can translate to earlier interventions, potential reductions in emergency presentations, and better patient education about disease trajectories. While the data suggest substantial improvements in detection sensitivity, clinicians must weigh the benefits against a potential rise in false positives and the resulting downstream testing. The consensus across credible sources emphasizes the need for clear risk communication with patients, explicit documentation of AI recommendations, and ongoing monitoring of how AI-assisted screening affects downstream care pathways. The evolving consensus reflects a broader trend toward blended human-AI decision-making in cardiovascular screening. (newsroom.heart.org)
Implementation timeline: aiming for scalable deployment
A practical pathway toward scale starts with pilot expansion to diverse clinical settings, accompanied by careful data governance and clinician training. Steps include (a) standardizing data capture protocols for audio quality, (b) integrating AI outputs into the clinic’s EHR or decision-support system, (c) establishing follow-up imaging criteria to manage false positives, and (d) monitoring impact metrics such as time to diagnosis, referral rates, and patient outcomes. The evidence across studies supports a staged approach: validate in multiple sites, confirm through blinded follow-up, then extend to broader populations with ongoing performance audits. The timing aligns with contemporary regulatory and professional expectations for digital health tools, including transparency about model performance and clinically meaningful outcomes. (newsroom.heart.org)
The Results
Increased detection sensitivity and improved case-finding
Across major studies, AI-enabled digital stethoscopes consistently outperformed traditional auscultation in detecting valvular heart disease. In the 2023 AHA presentation, the AI method achieved 94.1% sensitivity for detecting valvular disease versus 41.2% with the standard stethoscope. That improvement translated into identifying 22 previously undiagnosed cases among 369 participants, compared with 8 undiagnosed cases identified by conventional auscultation. The contrast is stark: AI-assisted screening not only catches more true positives but also does so in a way that is testable and verifiable through echocardiography as the reference standard. The implications for patient outcomes and health-system efficiency are profound, because earlier detection enables timely management before symptoms escalate. These numbers live in the documentation of the study and provide a benchmark for future comparisons. The study’s limitations—such as sample size and generalizability to broader populations—are noted by the authors, but the signal remains compelling. (newsroom.heart.org)
In a subsequent, real-world study reported by the European Heart Journal - Digital Health, the AI-enabled stethoscope demonstrated 92.3% sensitivity for detecting moderate-to-severe valvular disease, more than doubling the sensitivity observed with traditional listening (46.2%). This particular study was conducted in a real clinical setting with 357 patients aged 50 and above, reinforcing the external validity of the approach and its applicability outside tightly controlled research environments. While specificity showed a slight decline in some configurations, the authors and external commentators highlighted the clinical trade-off: the cost of some false positives may be acceptable when early pathology is unmasked, given the potential to avert adverse events through early intervention. The message to practitioners is clear: AI-enabled auscultation can materially raise the odds of catching significant valve pathology earlier, without replacing the clinical acumen of the physician. (escardio.org)
Earlier detection translates into actionable next steps
Beyond mere detection, the practical value lies in how AI-enabled auscultation channels patients toward timely echocardiography and intervention. The AHA study explicitly linked AI detection to an increased number of individuals being flagged for follow-up imaging within a short window (1–2 weeks). This alignment between screening and confirmatory testing helps close the diagnostic loop, reducing the window of uncertainty for patients and clinicians alike. Early diagnosis has downstream implications for treatment planning, surveillance, and risk stratification, potentially decreasing the likelihood of late-stage presentations and urgent admissions. In clinical storytelling from the field, physicians noted that AI-assisted sessions could improve patient engagement during the appointment, partially because patients could visually and audibly connect with the data underpinning the clinician’s assessment. These qualitative signals complement the quantitative improvements in accuracy and show the biopsychosocial benefits of AI-supported care. (newsroom.heart.org)
Performance in larger, real-world deployments and ongoing validation
The most recent, peer-reviewed and conference-linked results presented in 2026 reinforce the viability of AI-enabled stethoscope technology in routine practice. The ESC press release describes a single-arm, single-blinded prospective study involving 357 patients across three primary care settings, with the AI-enabled device achieving 92.3% sensitivity versus 46.2% for the traditional stethoscope. This real-world evidence bolsters confidence that the technology can perform in typical clinical workflows, not just in idealized research environments. A key caveat remains the trade-off with specificity, which saw a minor decline in some configurations. The take-home message for practitioners is nuanced: AI-enhanced screening may increase the detection of significant valves disease at the cost of a modest uptick in false positives, a balance many clinicians are prepared to manage when the downstream benefits include earlier treatment and reduced adverse events. As always, broader deployment will depend on local clinical guidelines, resource availability, and patient-centered communication strategies. (escardio.org)
Real-world engagement and patient experience
Qualitative observations from some reports indicate that patients respond positively when clinicians explain AI-supported findings in an accessible way. The Guardian’s coverage of UK-based work suggests that patient engagement—seeing and hearing data during the visit—might improve trust and adherence to recommended follow-up care. While this is a descriptive, media-grade view, it aligns with the broader emphasis on patient-centered communication when AI tools influence care decisions. High engagement can translate into better adherence to surveillance plans, timelier imaging, and a more proactive stance on health management—critical factors in chronic disease contexts where early intervention matters. The patient experience dimension adds a practical layer to the metrics, signaling potential reputational and satisfaction benefits for clinics that adopt AI-augmented auscultation thoughtfully. (theguardian.com)
Summary metrics in a cross-study view
- Sensitivity improvements: 41.2% (traditional) vs 94.1% (AI-enabled) in the 2023 AHA data; and 46.2% (traditional) vs 92.3% (AI-enabled) in the 2026 ESC study. Both studies indicate substantial gains in detecting valvular disease when AI analyzes digital stethoscope data. (newsroom.heart.org)
- Undiagnosed cases uncovered: 22 cases vs 8 cases in the AHA 2023 cohort, illustrating practical case-finding gains when AI augments auscultation. (newsroom.heart.org)
- Follow-up imaging: 1–2 weeks after the initial exam to confirm VHD status, underpinning a tighter diagnostic loop in AI-supported workflows. (newsroom.heart.org)
- Real-world multicenter evidence: 357 patients in the 2026 ESC study with robust multicenter data supporting AI performance in everyday clinic settings. (escardio.org)
- Specificity considerations: A modest trade-off in some configurations, highlighting the need for balanced interpretation and diagnostic pathways that minimize unnecessary testing. (newsroom.heart.org)
- Patient engagement signals: Anecdotal but positive reports on how AI-assisted visits can improve patient understanding and engagement with the diagnostic process. (theguardian.com)
Key Learnings
What worked well

- Substantial gains in sensitivity without sacrificing clinical judgment: Across credible studies, AI-enabled stethoscopes markedly improved the detection of valvular disease compared with traditional listening. This suggests that AI can uncover subtle acoustic patterns that human ears may miss, enabling clinicians to identify at-risk patients earlier and direct them to echocardiography or specialist care sooner. The combination of digital auscultation and AI created a more reliable screening signal in primary care, triaging patients who would otherwise fall through the cracks in routine exams. As one lead author noted during dissemination, AI-assisted screening can empower clinicians to screen more patients effectively and with confidence. (newsroom.heart.org)
- Real-world feasibility demonstrated in multicenter settings: The ESC 2026 study shows that AI stethoscope technology can perform in real-world clinic environments across multiple sites, an important milestone for scalability and adoption. The practical validation at three primary care sites suggests that integration with existing workflows is feasible without requiring overhauls of clinical infrastructure. This is a key driver for health systems considering investment in digital auscultation for routine screening. (escardio.org)
- Early case-finding translates to earlier actionable steps: The data demonstrate that AI-assisted screening can funnel patients toward echocardiography earlier in the disease course, which aligns with best practices for managing valvular disease and potentially improves outcomes by enabling timely intervention. This “screen-and-refer” approach resonates with clinicians who want to balance diagnostic thoroughness with resource stewardship. (newsroom.heart.org)
What didn’t go as smoothly
- Specificity trade-offs and false positives: A consistent caveat across studies is that increasing sensitivity with AI can be associated with a modest decline in specificity. In practice, this means more patients may undergo echocardiography or further testing than would occur with standard auscultation alone. While early detection is valuable, health systems must design follow-up pathways that minimize unnecessary testing and patient anxiety while preserving the benefits of early diagnosis. Ongoing refinement of AI models and clinician-guided thresholds will be necessary to optimize this balance. (newsroom.heart.org)
- Generalizability across diverse populations: Some study populations were specific (e.g., adults 50+, urban clinics, etc.), and investigators emphasize the need to validate AI models across broader demographic and geographic groups to ensure consistent performance. As adoption scales, attention to bias, data diversity, and local prevalence will be crucial to sustaining accuracy and fairness in screening outcomes. (newsroom.heart.org)
Lessons for scaling
- Align AI outputs with clear clinical pathways: The technology is a tool to augment, not replace, clinical reasoning. Successful scale requires well-defined workflows that specify when to order echocardiography, how to communicate AI results to patients, and how to monitor subsequent outcomes. This includes designing alerting rules, documentation templates, and follow-up protocols that make AI insights actionable and safe. (newsroom.heart.org)
- Invest in governance, privacy, and transparency: As digital auscultation becomes more widespread, health systems should invest in governance frameworks that address data privacy, model explainability, and ongoing monitoring of performance. Clinicians should have access to context about how AI decisions are made and should retain final responsibility for patient care decisions. The literature consistently highlights these concerns as essential components of responsible AI deployment in cardiology. (newsroom.heart.org)
- Prioritize patient-centered communication: Early data hint that patients respond positively to AI-assisted processes when they can see or hear the data driving recommendations. Transparent communication about what an AI reading means, what steps will follow, and why those steps are necessary can improve trust and adherence to follow-up plans. This social dimension matters as much as the technical performance in successful adoption. (theguardian.com)
Closing
The Cambridge Review’s investigation into AI stethoscope heart valve disease detection reveals a compelling narrative: digital auscultation paired with AI can meaningfully improve detection of valvular disease in real-world clinical settings, accelerating the diagnostic journey from screening to echocardiography and potential treatment. The data—from 94.1% sensitivity in the 2023 AHA study to 92.3% sensitivity in the 2026 ESC study—paint a consistent picture of a tool that helps clinicians see cases they might miss with traditional listening alone. Of course, the story remains nuanced. Trade-offs in specificity, the need for broader population validation, and the critical requirement for thoughtful workflow integration must guide deployment decisions. Still, the trajectory is clear: AI-enabled digital stethoscopes are moving from prototype to practical, scalable screening aids that can change how heart valve disease is detected and managed at the community level. The question now is not merely “Can AI detect more?” but “How can health systems deploy AI stethoscope heart valve disease detection responsibly, at scale, and in a way that meaningfully reduces patient risk and improves outcomes over time?” The evidence to date suggests a promising step in that direction, with ongoing research, thoughtful implementation, and a commitment to patient-centered care guiding the path forward. (newsroom.heart.org)